
Alginate Bead, Drug Delivery
Summary
Alginate beads are a hydrogel bead with a liquid center. They can be used as a drug delivery model, by loading them with a dye that diffuses out over time. This module focuses on both product and process engineering, two staples of chemical engineering. A system for the automated production of these beads is designed and created by students, and bead geometries and mass transfer properties are then analyzed.
General Information
Bibliography
- Farrell, Stephanie, and Jennifer Vernengo. “A Controlled Drug-Delivery Experiment Using Alginate Beads.” Chemical Engineering Education 46.2 (2012): 97-109. Print.
- Ritger, Philip L. and Peppas, Nikolaos A. “A simple equation for description of solute release I. Fickian and non-fickian release from non-swellable devices in the form of slabs, spheres, cylinders or discs.” Journal of Controlled Release, Volume 5, Issue 1, June 1987, Pages 23-36
Materials & Methods
In large part, this module is based on an article from Stephanie Farrell, and Jennifer Vernengo, referenced in the bibliography.
Required Items:
- Calcium Chloride – You can get this from any chemical supplier, or you can buy it cheaply as it is commonly used as ice melt (e.g.).
- Algenic Acid – You can get this from most chemical suppliers, such as Fisher Scientific or Sigma. Alternatively, you can purchase food grade alginate and CaCl2 and make edible beads (can be found on Amazon.com)…
- Food Coloring or Some Other Dye – This is needed only for the Mass Transfer portion of this module.
- Containers – Different shapes and sizes of inexpensive plastic containers to use as the main body of the apparatus. Two-liter bottles and other food-product containers work nicely.
- Pumps – They are used to move the two solutions. Water fountain (e.g.) and aquarium pumps (e.g.) are fairly cheap.
- Tubing – Make sure to have the correct tubing for the pumps to prevent leaks; a local home improvement store should have what you need.
- Plastic Fittings – These can be used to connect the tubing together and split flow lines. Such fittings can be found at a local home improvement store or online (e.g.).
- Spectrophotometer (optional) – Only needed if you will be conducting the mass transfer portion of this module. This is used to determine the rate of diffusion out of the beads. It is best to have several if there are a lot of groups. Instructions on how to make a cheap alternative are found here.
- Python 3.x (optional) – This is only needed for this module if you wish to use our program and an image to calculate bead diameters and eccentricities. This software can also be used to read from the spectrophotometer. Our Python scripts for this work will be given at the end of this module.
- Digital Camera – This is used to take an image of beads to be processed by our MATLAB program, or by hand using some other image software, such as photoshop. The higher the resolution the better.
- Filter Materials – Some way to separate the beads from the calcium chloride. Screens typically used on screen doors work well.
Note: It is best to have a wide variety of containers, fittings, tubing, filters, and pumps, as it increases the design possibilities.
Additional Helpful Tools:
- Tape, Hot Glue, Super Glue, Scissors, Dremel with an assortment of tools.
Instructions: Process Design:
First, mix the two required solutions in water (DI water is preferred but not necessary).
- 10 g/L Alginic Acid – About 20 mL per student team.
- 6 wt% CaCl2 – About 500 mL per team. Note that the concentration of salt may be treated as a design factor as well. There is a trade-off; both low and high salt concentration can lead to gel deformations.
In general, alginate beads are created by dripping the alginic acid into the calcium chloride solution, transforming the outer shell of the droplet into a hydrogel. The resulting beads must then be separated from the calcium chloride and loaded with a dye.
The design goals given to students for this project are to create an automated system which produces uniform, spherical alginate beads with a specified size (around 3 mm) which translates into a specific release rate. The student’s process must be able to convert 5 mL of alginic acid solution into beads continuously without interference, separate out the resulting beads, and recycle the calcium chloride solution.
This teaching module is meant to be a very open-ended design project in which the students are able to be create a wide variety of processes of their own making. We explain the basic chemistry to the students (as described in the background section of this module), show them an example formation of alginate hydrogel from the two solutions, and provide students with the chemicals, pumps, tubing, containers, dye, and mesh. A variety of each piece of equipment is offered and the students may request additional equipment.
Ultimately students end up with some process with a reaction process a separation process, and a recycle stream.


But each of these parts can result in a wide variety of student-designed processes, as seen below.


To load the beads we place them in a 5e-5 M solution of Malachite Green or fuchsin over a week, though any food coloring will work and the beads may be loaded much faster than 7 days. Alternatively, the dye may be placed in both the alginate and CaCl2 solutions and thereby the beads will be loaded at their creation.
Product Analysis:

After their beads are created and loaded, students may quickly determine size and eccentricity distributions using MATLAB’s image processing toolbox and a webcam (MATLAB Code links above). This program automatically takes images, measures the beads, and gives the students their size distribution and eccentricity. Alternatively, students may take an image of their beads with any digital camera and measure their diameters in pixels with a number of other image processing tools, such as Photoshop or imageJ. It is helpful to take the picture with as high of a resolution as possible and with all the beads spread out on a contrasting background (see the adjacent picture). Also, the background should not absorb the dye; we use wax paper. An object of known diameter should also be in the picture so that students can convert from pixels to millimeters; we use a poker chip.
Background for Teachers
The creation and analysis of alginate beads has been found to be effective in teaching concepts of mass transfer [1].
Hydrogel Formation
The hydrogel formation in this module is a product of the reaction between alginic acid and a calcium chloride solution. When dissolving calcium chloride in water, it dissociates and turns into positively charged calcium ions and negatively charged chlorine ions:

![]()
Alginic acid is a block copolymer consisting of two monomers: (1-4)-linked β-D-mannuronate and C-5 epimer α-L-guluronate. A block copolymer is a chain of monomers, there are sections of each monomer alone, and sections where the monomers alternate. Because calcium ions have a +2 charge, they are able to attract two alginic acid strands, linking them together. This new linked structure, though mainly water, now has some support and solidity.
In order to create a bead with a hydrogel shell and a liquid center the alginic acid is dripped into the calcium chloride. The calcium ions react with the alginic acid on the outside of the droplet first, then slowly diffuse into the droplet. Because of this, the longer the bead remains in the calcium chloride solution, the thicker the hydrogel shell will be. A thicker shell increases the amount of time it takes for the dye to diffuse into and out of the bead.
Mass Transfer
After the beads have been loaded with a dye they are placed into a beaker of water to test their release rate. Initially, the concentration of the malachite green inside the alginate beads is very high, and outside is low. According to Fick’s first law the molecules tend to move from high to low concentration. Fick’s first law states that

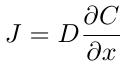
where J is the flux of the molecules, which is defined as the movement of molecules across a unit area per unit time. This equation states that the flux of a diffusing species is proportional to the change concentration, C, over distance, x. Here D is the diffusion coefficient, which depends on the diffusing species. However this equation is too simplified to use as it was derived for diffusion in one dimension, and there is a non-uniform resistance to flow due to the shell of the alginate bead. Because the hydrogel shell is very porous on the molecular level, the dye is able to slowly flow through it.
The general diffusion equation in spherical coordinates is:

![]()
This equation may seem very complicated, but it can be simplified to account for spheres that only have a concentration gradient in the radial direction.

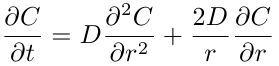
As this is still more complicated than we would like to work with, we can use a simpler empirical relationship. One empirical relationship for the diffusion from a non-swellable spherical polymer is proposed in [2]:

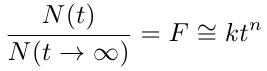
where F is the fraction of the dye that has diffused, and N(t) is the total number of moles released at time t. The data may be fit to this equation and the constants n and k can be estimated. The constant n should range from 0.43 to 0.5 depending on the beads’ eccentricities, where 0.43 corresponds to a perfectly spherical bead and 0.5 corresponds to a flat membrane.
Note: It is important to remember that this model is only an approximation. It is impossible for the fraction of the dye that has diffused out to become greater than 1, contrary to this empirical model. As a rule of thumb, this equation should not be used for values of F > 0.6.
More detailed theory and a simulation of this experiment may be found here:
Instructional Procedures
- Students are shown the reaction of alginic acid and calcium chloride and are told the goals of the project.
- Each group is given an assortment of materials including 5 mL of alginic acid.
- The groups are told to make a 10% wt solution of calcium chloride and to try to create some alginate beads by hand.
- Each group is then supposed to design and build an automated system that can continually convert 5 mL of alginic acid into alginate beads without interference. The system must also separate out the beads and recycle the calcium chloride solution.
- This project is meant to be a very open-ended design experiment. As such, little instruction on the design and construction of the apparatus is given to the students.
- Students then make enough calcium chloride solution for their system.
- Before turning on any pumps, students need to check off their design with the instructor to check for leaks and proper wiring.
- Groups then use 5 mL of alginic acid to create alginate beads. If only one day is allotted for the project, the dye should be mixed in the alginic acid before the beads are made.
- The beads should then be placed in a concentrated solution of malachite green in order to load the beads with the dye unless the dye was previously mixed in.
- Next, the beads need to be placed on a sheet of wax paper. Beads should be separated from each other and any water should be removed from the beads.
- Using a webcam and MATLAB, the distribution of the beads sizes and eccentricities is determined.
- The beads are then put in a stirred beaker of water. The concentration of dye in the water is measured with a spectrophotometer and recorded.
- Students determine the rate of diffusion of dye out of the beads and compare it to theory.
- Depending on the chosen goals for the project, students may need to alter some parameters of their apparatus in an attempt to make more spherical beads or match a specified diameter or release rate.
- Students may also use the online simulation of this experiment, found here: http://vstem.org/app/DiffusionSphere/
- to compare their data to theory or test out various scenarios.

![]()
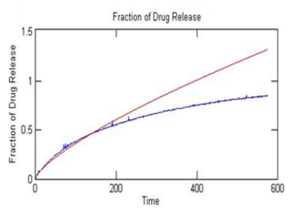
Then the rate of diffusion may be determined by tracking the amount of dye that diffuses out of the beads over time using a spectrophotometer. A known volume and mass of beads (~5 mL) are placed in 100 mL of DI water that is stirred continuously. Over time, the dye diffuses out of the beads and begins concentrating the water. Samples of the water may be taken periodically or continuously and measured in a spectrophotometer (Instructions on how to build one with a flow cell here) to determine dye concentration. Concentration should be measured as often as possible from the time beads are introduced to the water for at least 20 minutes. Concentrations should asymptotically reach a maximum over time, as seen in this example data:
This data may then be analyzed as described in the background area of this module
The students may go back and alter several parameters in order to create more uniform beads that are closer to the design goals. Some of the parameters that may be adjusted include the concentrations and flow rates of the alginate and calcium chloride solutions, the drip height and tip diameter, and the residence time of the beads in the calcium chloride solution.
Due to the number of goals and parameters that can be adjusted, this project may be adapted to various levels of difficulty. We have used this project for teaching freshman and senior students in the chemical engineering program, and for junior high and high school students as part of the Science Olympiad. The real-world applications of alginate beads make it attractive as a teaching module. According to a student survey in our chemical engineering freshman design laboratory, this was the most-enjoyed project out of seven, and it was also ranked the highest in the students’ assessment of which teaching modules were most educational.

Source Code